Imke
Visser

Project
Tags
Promotor
Marion Koopmans
Co-promoter(s)
Barry Rockx
Institute(s)
Erasmus MC
Description PhD project
The primary mode of transmission of arthropod-borne viruses is through the bite of an infected mosquito, where virus is injected into the host skin along with mosquito saliva. Although modulation of the host immune response by mosquito saliva alone or during viral infection has been studied, full evaluation of the complex dynamics between different viruses, mosquitoes, and the ecosystem is lacking. Specifically, the differential effects of variant combinations of vector and virus species on the modulation of the local (skin) and systemic immune response and outcome of pathogenesis in the vertebrate host has not yet been extensively studied. In addition, recent introduction of exotic mosquito species in the Netherlands, such as the Asian bush mosquito Aedes japonicus, warrants investigation of the ability of such mosquitoes to transmit emerging viruses to humans and cause disease. Therefore, examining the effect of a mosquito bite from different relevant mosquitoes under changing environmental influences is imperative. I want to study the early interactions between virus, vector (saliva), and the first organ that the virus encounters: the skin. Concurrently, I want to investigate whether these early interactions subsequently influence the host viremia and viral pathogenesis. Overall, I want to see whether different mosquito species trigger distinct host immunopathogenic responses, accounting for diverse clinical outcomes of viral infection.
Furthermore, alternative transmission routes of arthropod-borne viruses such as oral (TBEV) and respiratory (JEV) have been reported, but the importance of non-arthropod transmission during arthropod-borne disease outbreaks is not well-studied. Our approach is expected to identify key parameters that are instrumental in virus transmission and pathogenesis.
Research questions / objectives
- Identify the dose and route(s) (vector and non-vector routes) via which virus is most efficiently transmitted to the vertebrate host, and discern the effects of mosquito bite on the outcome of viral disease
- Determine the minimal level of host viremia contributing to maintaining a transmission cycle
- Elucidate the mechanisms by which mosquito saliva from different species differentially contributes to the transmission, enhancement, and dissemination of distinct viruses to and within the vertebrate host
- Determine whether and how anthropogenic stressors influence the ability of a mosquito to efficiently transmit virus to the vertebrate host and cause disease
Timeline
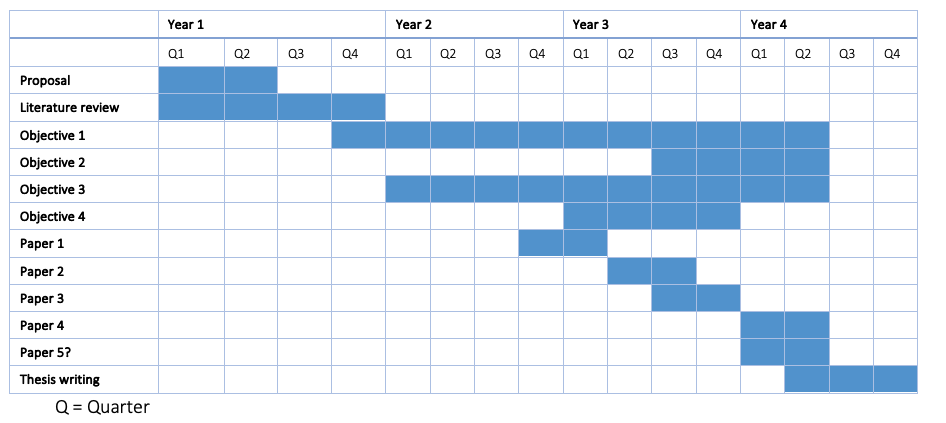
Tags matching with the contents of track 17
Methods
-
Labwork
- PCR: Virus detection in tissues and cell culture
- ELISA: Antibody detection upon in vivo virus infection with/without mosquito saliva
- RNA/DNA extraction: RNA extraction from tissue samples to determine viral load
- Virus neutralization: Perhaps after detecting antibody upon in vivo virus infection with/without mosquito saliva (see above), determine whether these antibodies are neutralizing
- In vivo: To identify the dose and route through which the vertebrate host is most efficiently infected; to elucidate the mechanisms by which mosquito saliva from different species differentially contributes to the transmission, enhancement, and dissemination of distinct viruses to and within the vertebrate host; to determine the minimal level of host viremia contributing to maintaining a transmission cycle; to investigate non-vector borne routes of viral infection
- In vitro: Develop in vitro models to investigate the mechanisms by which mosquito saliva from different species differentially contributes to the transmission, enhancement, and dissemination of distinct viruses to and within the vertebrate host using relevant (human and bird) cell lines
- Ex Vivo: Ex vivo skin tissue for viral infection experiments with/without mosquito saliva
- Plaque Assay: Either plaque assays or TCID50 will be used to determine viral titers of in vitro and in vivo infection experiments
- Living mosquito experiment: I may use mosquito saliva from mosquitoes reared under living lab conditions (collab. with Maarten Schrama, LU) if possible. I will use live (uninfected or experimentally infected) mosquitoes to be used for transmission studies (collab. with Sander Koenraadt/Gorben Pijlman, WUR)
- Living animal experiment: To identify the dose and route through which the vertebrate host is most efficiently infected; to elucidate the mechanisms by which mosquito saliva from different species differentially contributes to the transmission, enhancement, and dissemination of distinct viruses to and within the vertebrate host; to determine the minimal level of host viremia contributing to maintaining a transmission cycle; to investigate non-vector borne routes of viral infection
Modelling
- Epidemiological modelling: I will determine the minimal level of host viremia contributing to maintaining a transmission cycle, which will give insight into the spread of a virus through a population considering the amount of virus present in a patient needed for a biting mosquito to become infected and subsequently be able to infect other people
Literature
- Literature review: I am writing a literature review on the transmission of arthropod-borne viruses from vector to host and the effect of vector saliva on viral transmission, pathogenesis, and the host immune system
Topics
-
Vector:
- Vector movement patterns, migration: If a certain mosquito species enters Europe/the Netherlands it is interesting for my project to obtain some saliva from these species (dependent on the mosquito trappers, WUR?)
- Vector susceptibility: In my project I want to see whether a mosquito can become infected by biting an infected vertebrate (e.g. mouse) displaying different levels of viremia
- Vector competence: When virus is detected in the vector saliva (= labelled as competent vector) I will continue the vector competence research by determining whether the vector can indeed transmit the virus to a vertebrate host (using in vivo experiments)
Host:
- Host immune response: I will look into the local (skin) as well as systemic host immune response upon viral infection with or without vector saliva.
- Host susceptibility: I am going to investigate whether certain (human, bird) skin cells (in vitro) as well as whole vertebrates (in vivo) are susceptible to viral infection and whether the susceptibility and pathogenesis is influenced by vector saliva.
Virus:
- Pathogenicity: I am going to investigate whether certain (human, bird) skin cells (in vitro) as well as whole vertebrates (in vivo) are susceptible to viral infection and whether the susceptibility and pathogenesis is influenced by vector saliva.
- Virus-host interaction: I will look into the local (skin) as well as systemic host immune response upon viral infection with or without vector saliva.
Interventions:
- Vaccines: Using our transmission model (host + mosquito bite), a potential vaccine can be tested: vaccinating mice to subsequently infect (challenge) with a wildtype virus transmitted by a vector instead of a needle. This represents the most natural way of how people can become infected by an arthropod-borne virus.
- Preparedness: When an invasive mosquito species emerges in Europe / the Netherlands, we can combine (saliva from) these mosquito species with a range of viruses (either present or not yet present in Europe) and infect our mouse model to determine whether a potential disease outbreak could occur.
- Diagnostics: Samples from in vivo experiments can be used for validation of diagnostic assays.
- Other interventions: As for vaccines (see above), but with antivirals and neutralizing antibodies.
Species
-
Species:
- Human: All the in vivo and in vitro work will be mostly directed to human health, therefore mostly primary human tissue (in vitro, ex vivo) and a mouse model (in vivo) will be used.
- Bird: Experimental infection in birds (collab. with Gianfillipo, UU)
- Mosquito: I will obtain mosquito saliva from different mosquito species to determine their role in arbovirus transmission to the vertebrate host.
- Other arthropods: As under ‘mosquito’, but then I might also include tick saliva in my project.
Virus
-
Virus:
- Virus (general): We will carry out experiments on the priority arboviruses as decided by the OHPACT consortium. Currently the main viruses we wish to study are highlighted below
- West Nile virus: Co-ciruclating with USUV, circulating in Europe and recently detected the Netherlands. WNV infection is enhanced when co-inoculated with saliva from North-American mosquito species, but not much is known about the effect of European mosquito species saliva on the pathogenesis of European WNV isolates in the vertebrate host.
- Usutu Virus: As with WNV
- Japanese Encephalitis Virus: As with WNV (minus the co-circulation). In addition, JEV transmission also occurs via the respiratory route (pigs), and thus I can use JEV as a model virus for an airborne-transmitted arbovirus.
- Tick-Borne Encephalitis Virus: The geographical range of tick species is expanding, and oral infection of TBEV through the consumption of infected raw milk is occurring in Europe.
- Chikungunya Virus: The global spread and establishment of aegypti and Ae. albopictus has led to the emergence of CHIKV in (Southern) Europe. I want to investigate whether saliva from mosquito species in Northern Europe / the Netherlands influences the transmission of CHIKV to a vertebrate host.
- Mayaro Virus: MAYV circulates in tropical rainforests in Central and South America. However, it has been predicted (modelled) that MAYV will expand its range in the coming years. South-American aegypti and Ae. albopictus are competent vectors for MAYV, indicating the potential of MAYV to establish an urban transmission cycle. I want to investigate whether (saliva from) European mosquito species are able to transmit/potentiate MAYV to a vertebrate host.